As organizations move toward "Privacy by Design," assurance layers will become embedded infrastructure, ensuring that healthcare innovation continues without compromising patient trust
NEWARK, DE / ACCESS Newswire / April 1, 2026 / The global healthcare data de-identification assurance market is entering a high-growth phase, projected to expand from USD 0.5 billion in 2026 to USD 1.6 billion by 2036, registering a CAGR of 12.3%, according to Future Market Insights (FMI). Growth is being fueled by the rising need for verifiable data privacy, increasing use of healthcare data in AI and clinical research, and regulatory pressure for HIPAA-compliant anonymization.
Healthcare data de-identification assurance goes beyond basic data masking by providing statistical validation, risk scoring, and audit trails that ensure datasets carry minimal re-identification risk. As healthcare organizations increasingly monetize secondary data and participate in global research collaborations, the demand for provable, compliant anonymization has become mission-critical.
Get Access of Report Sample: https://www.futuremarketinsights.com/reports/sample/rep-gb-32504
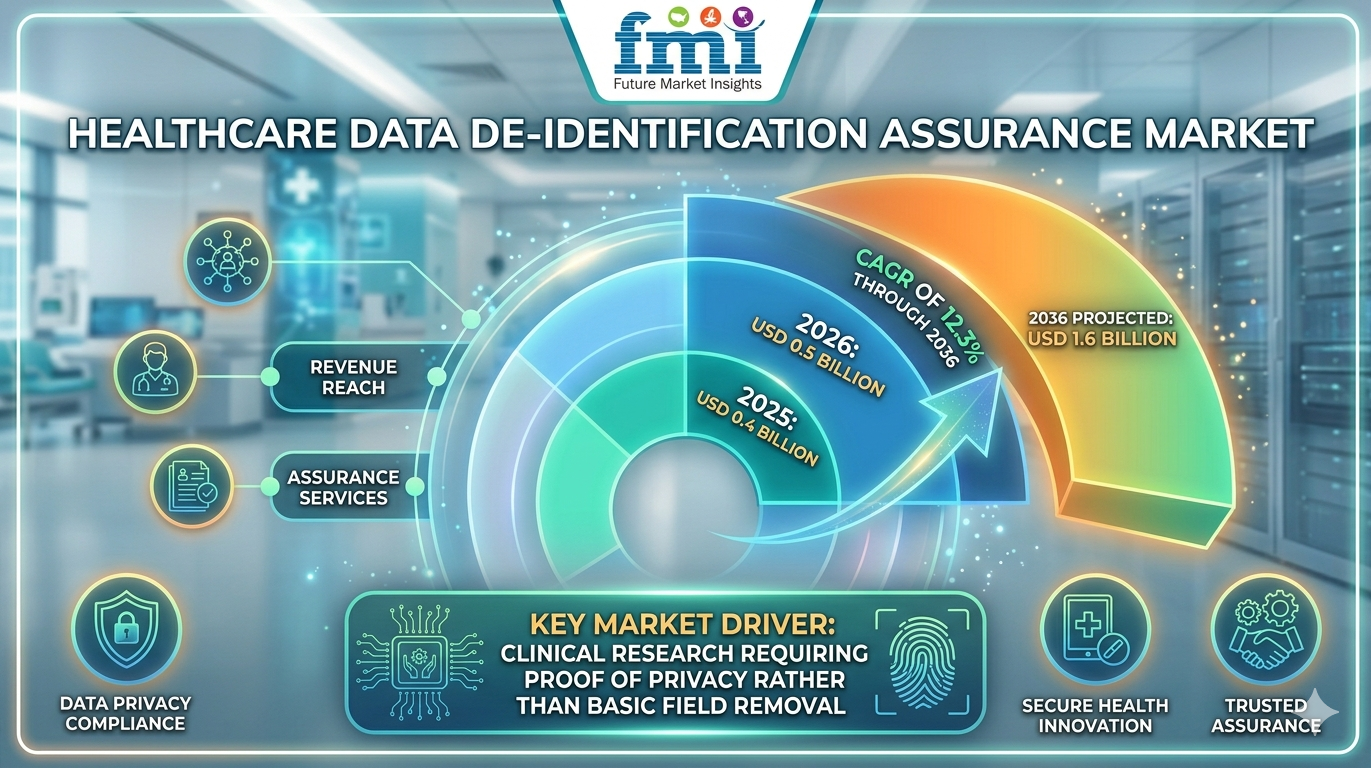
Healthcare Data De-Identification Assurance Market Quick Stats Snapshot
2026 Market Value: USD 0.5 Billion
2036 Forecast Value: USD 1.6 Billion
CAGR (2026-2036): 12.3%
Leading Technique: Expert Determination (38.0% share)
Top Deployment Mode: Cloud (44.0% share)
Leading Buyer Segment: Providers (32.0% share)
Key Growth Driver: Demand for verifiable privacy in AI and secondary data use
Innovation Focus: Continuous risk scoring, audit trails, and privacy engineering
Market Momentum: From Compliance to Continuous Privacy Assurance
The market is rapidly evolving from a compliance-driven function to a strategic enabler of data-driven healthcare. Organizations are shifting from one-time anonymization audits to continuous monitoring systems that dynamically assess re-identification risk as datasets evolve.
Key growth drivers include:
Rising demand for AI training datasets with privacy guarantees
Expansion of secondary data monetization models
Regulatory requirements for HIPAA expert determination and global data protection laws
Increased reliance on real-world evidence (RWE) in clinical research
Healthcare providers and life sciences firms are recognizing that failure to ensure robust de-identification can result in regulatory penalties and loss of data licensing revenue.
Segment Intelligence: Technology and Adoption Trends
Technique Leadership
Expert determination dominates with 38.0% share, driven by preference for statistical risk modeling over rule-based safe harbor methods
Tokenization and synthetic data are gaining traction for advanced privacy-preserving analytics
Hybrid approaches are emerging for balancing data utility and privacy
Data Type Insights
Structured data leads with 34.0% share due to EHR interoperability
Rapid growth in unstructured clinical text and imaging data, requiring advanced NLP and DICOM anonymization tools
Assurance Layer Evolution
Software-led solutions dominate (41.0% share) enabling real-time risk scoring
Audit evidence and monitoring tools are becoming essential for regulatory compliance
Expert services remain critical for complex or novel datasets
Regional Insights: Asia-Pacific and North America Drive Expansion
India: Fastest-growing market at 15.2% CAGR, driven by digital health initiatives and large-scale data generation
China:14.6% CAGR supported by stringent privacy regulations and national health data platforms
United States:13.0% CAGR due to HIPAA enforcement and advanced research ecosystems
Germany:12.5% CAGR backed by GDPR compliance and digital healthcare infrastructure
United Kingdom:12.3% CAGR driven by NHS data transformation programs
North America leads in adoption due to mature healthcare IT systems, while Asia-Pacific is emerging as a high-growth hub due to rapid digitization and regulatory evolution.
Competitive Landscape: Technology Platforms and Network Effects
The market is moderately consolidated at the technology level, with strong competition among platform providers and fragmented expert service vendors.
Leading Market Players Include:
Datavant
IQVIA Privacy Analytics
John Snow Labs
MDClone
Privacert
TripleBlind
Duality Technologies
Enveil
Key strategies include:
Building tokenization ecosystems and network effects
Expanding automated risk-scoring platforms
Offering end-to-end audit trails and compliance tools
Developing privacy-enhancing technologies (PETs)
Key Growth Drivers and Challenges
Drivers
Rising demand for privacy-preserving AI training data
Growth in clinical research and real-world evidence applications
Regulatory push for verifiable anonymization standards
Increasing secondary data licensing and monetization
Challenges
Trade-off between data utility and privacy protection
Complexity of multi-modal data de-identification (EHR + imaging + claims)
High implementation costs for advanced assurance platforms
Need for continuous validation and audit readiness
Strategic Takeaways for Decision Makers
Invest in continuous risk-scoring and monitoring platforms
Transition from manual audits to automated assurance layers
Prioritize expert determination and hybrid techniques
Build scalable cloud-based anonymization infrastructure
Align privacy strategies with AI, RWE, and data monetization goals
Why This Market Matters Now
The convergence of AI-driven healthcare, global data-sharing initiatives, and stringent privacy regulations is redefining how medical data is used and protected. Healthcare data de-identification assurance is no longer optional-it is foundational to enabling secure, scalable, and compliant data ecosystems.
As organizations move toward "Privacy by Design," assurance layers will become embedded infrastructure, ensuring that healthcare innovation continues without compromising patient trust.
For an in-depth analysis of evolving formulation trends and to access the complete strategic outlook for the Healthcare Data De-Identification Assurance Market through 2036, visit the official report page at: https://www.futuremarketinsights.com/reports/healthcare-data-de-identification-assurance-market
Explore More Related Studies Published by FMI Research:
Healthcare Regulatory Affairs Outsourcing Market: https://www.futuremarketinsights.com/reports/healthcare-regulatory-affairs-outsourcing-market
Healthcare Business Intelligence Market: https://www.futuremarketinsights.com/reports/healthcare-business-intelligence-market
Healthcare Air Purifier Market: https://www.futuremarketinsights.com/reports/healthcare-air-purifier-market
Healthcare AI Computer Vision Market: https://www.futuremarketinsights.com/reports/healthcare-ai-computer-vision-market
About Future Market Insights (FMI)
Future Market Insights (FMI) is a leading provider of market intelligence and consulting services, serving clients in over 150 countries. Headquartered in Delaware, USA, with a global delivery center in India and offices in the UK and UAE, FMI delivers actionable insights to businesses across industries including automotive, technology, consumer products, manufacturing, energy, and chemicals.
An ESOMAR-certified research organization, FMI provides custom and syndicated market reports and consulting services, supporting both Fortune 1,000 companies and SMEs. Its team of 300+ experienced analysts ensures credible, data-driven insights to help clients navigate global markets and identify growth opportunities.
For Press & Corporate Inquiries
Rahul Singh
AVP - Marketing and Growth Strategy
Future Market Insights, Inc.
+91 8600020075
For Sales - sales@futuremarketinsights.com
For Media - Rahul.singh@futuremarketinsights.com
For web - https://www.futuremarketinsights.com/
SOURCE: Future Market Insights, Inc.
View the original press release on ACCESS Newswire




